Orchestra Life Sciences’ Year-End Perspectives
The life sciences sector entered 2025 with considerable momentum and concluded the year having experienced meaningful scientific, regulatory, and operational developments. Demand for advanced therapies continued to increase, global supply chains adjusted to evolving geopolitical and commercial conditions, and regulatory pathways evolved in ways that influenced development and commercialization strategies. Radiopharmaceuticals advanced further into the commercial landscape, and in vivo gene editing demonstrated continued progress within select clinical research programs.
For Orchestra Life Sciences, these developments reflect ongoing changes in how organizations will design, construct, and manage the technical and regulatory infrastructure required to support next-generation therapeutics. Across cell therapy, gene therapy, radiopharmaceuticals, and nuclear medicine, 2025 contributed to a foundation that will shape therapeutic innovation in the years ahead.
Manufacturing Investment Trends and Capacity Expansion
In the first half of 2025, more than $158 billion in US-based manufacturing investments were announced, underscoring a sustained commitment to domestic capacity expansion across life sciences. This investment wave has more immediate execution implications that are now shaping project outcomes.
In practice, the pace and scale of these commitments are forcing companies to lock critical facility decisions much earlier than in prior expansion cycles. Utility capacity, cleanroom classification, radiation shielding, and construction sequencing increasingly must be defined upstream to avoid downstream redesign, schedule compression, and cost escalation. Recent announcements from leading biopharmaceutical manufacturers illustrate not only the breadth of modalities being supported, but also the growing complexity of facilities expected to serve biologics, CGT platforms, radiopharmaceuticals, and hybrid pipelines within a single infrastructure footprint.
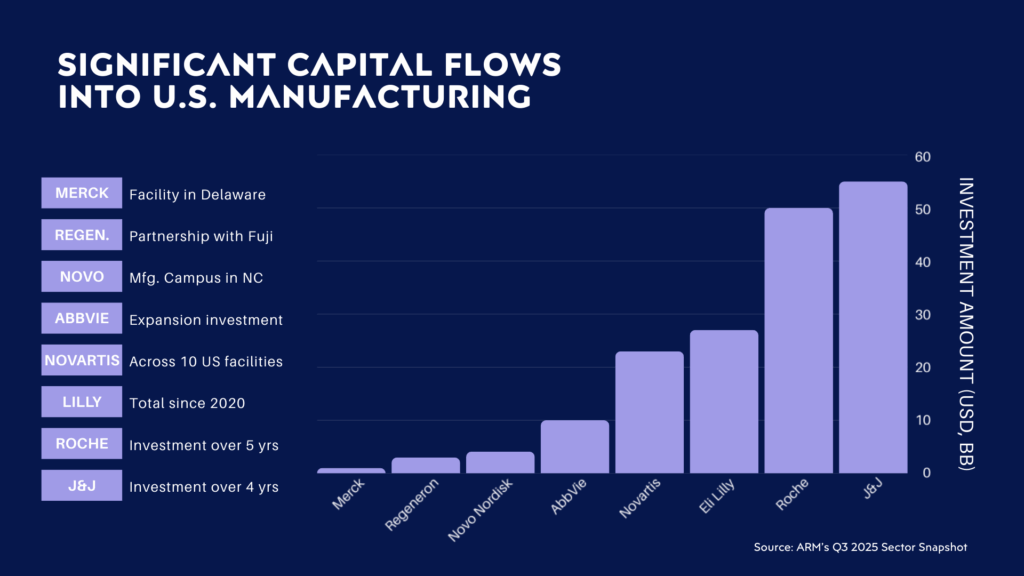
Industry analyses, including ARM’s Q3 2025 Sector Snapshot, reinforce that onshoring is no longer a single-modality exercise. As pipelines diversify, execution risk shifts from capital availability to design realism, cross-modal integration, and early risk control. Facilities that are not designed with regulatory, operational, and lifecycle constraints in mind from the outset face delayed startups and limited flexibility once assets are built.
Regulatory Acceleration, Platform Designations, and CAR-T Access Changes
Regulatory activity in 2025 demonstrated continued efforts to enhance the efficiency and clarity of pathways supporting advanced therapies. According to ARM’s Q3 2025 Sector Snapshot, while the cell and gene therapy sector in the United States is only on pace to see 4-5 approvals in 2025, next year is shaping up to see a rebound with four PDUFA dates already scheduled in H1 2026. The US pipeline remains active, with at least seven companies aiming to submit their regulatory applications in 2026.
These patterns are consistent with the continued maturation of the clinical pipeline and the FDA’s established use of expedited programs for products addressing unmet needs. The agency continued to apply platform-related regulatory approaches intended to support more efficient CMC review across related submissions
In June, the FDA removed REMS requirements for currently approved BCMA- and CD19-directed autologous CAR-T therapies. This adjustment had implications for operational feasibility and patient access by lowering some of the logistical requirements that historically limited broader utilization.
Advancing Approvals and Pipeline Momentum in CGT and In Vivo Editing
Clinical activity in 2025 reflected continued momentum across cell and gene therapy and in vivo gene editing programs, with FDA actions signaling a more mature and differentiated regulatory landscape. As reported in ARM’s Q1 2025 Sector Snapshot, non-viral delivery systems and genome-engineering platforms are increasingly advancing into human studies, expanding the breadth of modalities entering clinical development.

While these signals suggest greater regulatory clarity, they do not reduce the execution bar. As CGT programs move toward pivotal stages, facilities must still demonstrate validated processes, trained operators, and inspection-ready systems from day one. Scale-out introduces additional operational pressure, particularly on QC throughput, material flow, and workforce availability where insufficient modeling or lack of redundancy can quickly become bottlenecks as programs transition from clinical to commercial intent.
In parallel, progress in in vivo gene editing is shifting manufacturing and CMC risk earlier in development. Vector sourcing strategies, raw material qualification, and comparability planning now surface sooner, often before facility and supply chain assumptions are fully defined. Without early alignment across CMC strategy, sourcing, and operational readiness, these risks can compound as programs advance.
Together, these dynamics reinforce a consistent execution lesson across CGT and in vivo platforms: regulatory engagement may evolve, but success continues to depend on early readiness, disciplined scale planning, and infrastructure built to withstand inspection, expansion, and complexity, not just initial clinical entry.
Radiopharmaceuticals & Nuclear Medicine: Infrastructure Comes Into Focus
Radiopharmaceuticals continued their advance toward commercialization in 2025, with organizations increasing investment in production capacity, isotope supply, and enabling infrastructure. As programs move beyond clinical proof-of-concept, the focus has shifted from scientific feasibility to whether facilities and operating models can reliably support commercial execution.
At the facility level, this transition exposes a critical execution reality: FDA sterility expectations and NRC radiation safety requirements converge directly on design decisions. Hot cell configuration, ventilation zoning, pressure cascades, and exhaust handling all materially affect throughput, inspection readiness, and long-term operational flexibility. Facilities that treat these requirements sequentially rather than in an integrated manner face rework, constrained operations, or delayed startup.
In parallel, growing demand for radioligand therapies is driving more distributed manufacturing and supply models, increasing coordination risk across sites, vendors, and logistics partners. As supply networks expand, execution risk shifts from individual assets to end-to-end alignment, where late missteps in vendor integration, material flow, or release timing can delay patient access despite available capacity.
Across Orchestra engagements, similar themes have emerged. Organizations are emphasizing GMP-compliant radiopharmaceutical facilities, resilient supply chain design, and execution models capable of meeting FDA and NRC expectations as the modality progresses.
What This Year Means for Orchestra Life Sciences
Throughout 2025, Orchestra Life Sciences supported clients navigating accelerated scientific progress and evolving regulatory and manufacturing requirements. Our work included:
- Leading capital project strategies for radiopharma, CGT, and biologics programs with compressed timelines
- Supporting supply chain planning across Pb-212, Ac-225, and Lu-177 initiatives
- Preparing facilities for GMP inspection and clinical production through readiness and operational excellence programs
- Advancing workforce development initiatives for nuclear medicine, radiopharmacy, and advanced therapies
The developments observed in 2025 demonstrate continued integration of scientific innovation, regulatory expectations, and manufacturing strategy across advanced modalities. As the field enters 2026, organizations that invest in infrastructure, execution capability, and strategic preparedness may be better positioned to adapt to the evolving therapeutic landscape.
Orchestra Life Sciences remains committed to supporting this progression and to contributing the expertise necessary to bring next-generation therapies to patients.

