Workforce Readiness as a Strategic Determinant of Growth
The life sciences industry has entered a phase where scientific acceleration must be matched by operational maturity. Radiopharmaceutical pipelines are expanding rapidly, cell and gene therapies are moving toward commercial scale, and precision medicine continues to reshape expectations around targeted treatment.
Capital investment remains strong. Yet a structural constraint has become increasingly apparent: the availability of a workforce prepared to execute in highly regulated, technically complex manufacturing environments.
A Q3 2025 Area Development analysis captured this shift directly, observing that “a state-of-the-art facility without the skilled workforce to run it is simply a stranded asset.” As companies evaluate locations for new GMP facilities, workforce strategy has emerged as a decisive factor in site selection decisions.

The Radiopharmaceutical Talent Crunch
The urgency of this issue is particularly evident in radiopharmaceuticals.
In February 2026, BioSpace reported that escalating investment from AstraZeneca, Bristol Myers Squibb, Eli Lilly, and Novartis has intensified competition for professionals capable of operating at the intersection of nuclear science, pharmaceutical GMP, and regulatory compliance.
Radiopharmaceutical manufacturing operates under dual oversight from FDA GMP requirements and the Nuclear Regulatory Commission. This “double-regulated” environment demands a rare combination of competencies:
- Radiochemistry expertise
- Nuclear engineering knowledge
- Radiation safety fluency
- GMP documentation discipline
- Regulatory alignment across agencies
Few professionals are trained comprehensively at this intersection. As development pipelines expand, the execution gap continues to widen.
Workforce as a Technical Discipline
The industry has begun to formally recognize workforce development as a technical pillar rather than an auxiliary function.
In January 2026, ISPE announced the establishment of its Radiopharmaceuticals Community of Practice, identifying education and workforce development as defined technical pillars. This reinforces that training underpins:
- Engineering execution
- Quality assurance and validation
- Supply chain resilience
- Radiation safety compliance
- Technology transfer readiness
Many professionals at Orchestra Life Sciences are active ISPE members. Through that engagement, we see firsthand that workforce readiness must be integrated into facility planning, GMP startup strategy, and regulatory preparation from the start.
Philadelphia: From Incentives to Intentional Talent Strategy
The workforce conversation is also reshaping regional competitiveness.
Greater Philadelphia has positioned itself as a workforce-forward life sciences ecosystem, particularly in cell and gene therapy manufacturing and increasingly in radiopharmaceutical development.
The region’s foundation includes:
- A concentration of cell and gene therapy manufacturing operations
- Expanding radiopharmaceutical activity
- Academic institutions aligned with biotechnology and regulatory sciences
- Workforce organizations focused on accelerated technical career pathways
- Structured employer–education partnerships designed to anticipate skill gaps
This represents a broader shift from incentive-driven growth to intentional talent strategy. Long-term competitiveness depends on cultivating and sustaining specialized workforce capabilities aligned with emerging modalities.
Orchestra’s Commitment to Human Infrastructure
Orchestra Life Sciences relocated its headquarters to the Philadelphia Navy Yard with this ecosystem perspective in mind. The decision reflects a recognition that technical consulting, facility execution, and workforce development are interdependent components of a resilient advanced therapies cluster.
In 2026, Orchestra will launch a specialized workforce development platform co-located with our Navy Yard operations. The objective is execution-aligned preparation for GMP-regulated advanced therapy manufacturing.
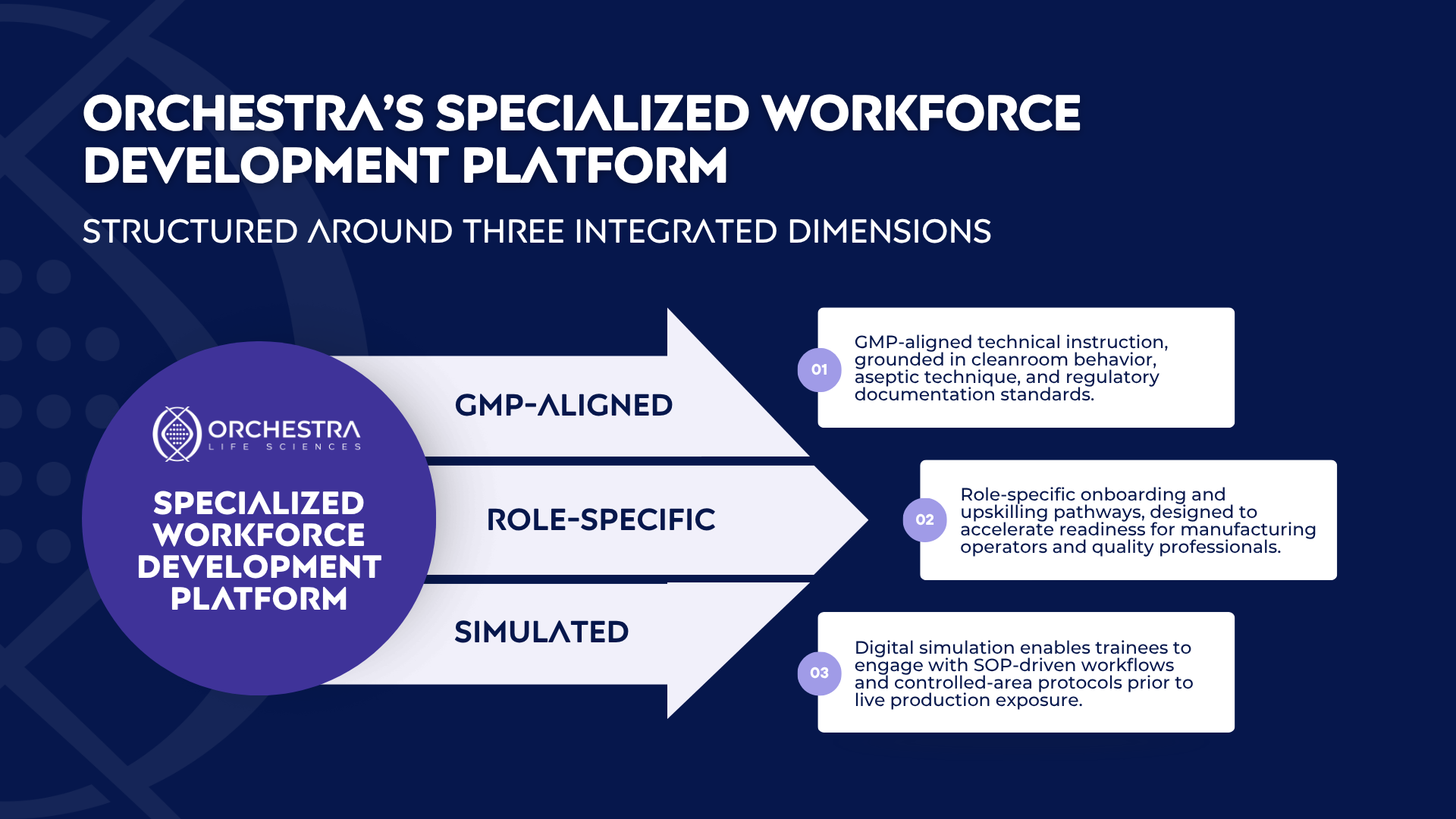
Our guiding principle is that structured experience must precede operational responsibility. By replicating real-world conditions within a controlled training framework, organizations can reduce onboarding variability and reinforce compliance discipline.
Building Competitive Advantage Through People
The next generation of life sciences hubs will distinguish themselves not by facilities alone, but by their capacity to cultivate specialized and adaptable talent.
Sustainable competitive advantage will depend on:
- Talent density in specialized GMP and quality roles
- Workforce adaptability across emerging modalities
- Structured cross-sector upskilling pathways
- Regulatory fluency in complex, dual-oversight environments
- Execution maturity at clinical and commercial scale
Advanced therapies ultimately reach patients through disciplined manufacturing and regulatory rigor. Those outcomes depend on people prepared to operate within highly controlled environments.
At Orchestra Life Sciences, we view workforce development as a long-term investment in execution excellence for our clients, for the ecosystem, and ultimately for the patients these therapies are designed to serve.

